New nanoparticle cancer therapy takes a step closer to clinical trials
A tumour-promoting protein, (SK1), was identified in prostate and breast cancer cells which reduces the effectiveness of chemotherapy.
Based on the above, a novel drug delivery system was designed using nanoparticles containing a combination of clinically approved SK1 inhibitor Fingolimod together with Taxane chemotherapy. This unique system targets cancer sites with greater accuracy - safely delivering powerful drugs and increasing the efficiency of the chemotherapy treatment.
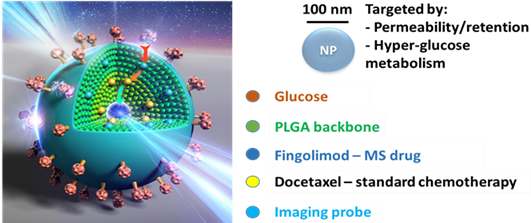
UEA researchers believe the new drug delivery system, once fully developed, will result in more positive outcomes in prostate and breast cancer patients.
This EIRA-funded proof of concept project enabled the novel cancer treatment to move closer towards human phase 1 clinical trials.
The Challenge
Taxane chemotherapy is offered to patients with advanced or metastatic cancers, but has limited effect due to its dose-limiting toxicity, side effects and the emergence of chemoresistance. i.e. tumours developing a resistance to the drug over time.
UEA researchers studying chemoresistance found a protein (SK1) was linked to the problem. Fingolimod is a clinically approved drug which suppresses the activity of SK1 and therefore a potentially useful drug to combat chemoresistance. However, as it supresses the immune system it cannot currently be used as a cancer therapy.
The challenge was therefore to develop a new delivery system for the drug treatment.
The Approach
An enzyme called SK1 was identified in prostate and breast cancer cells. When studied it was found that if this enzyme’s activity was suppressed then cancer cells became more susceptible to Taxane chemotherapy. Building on this finding it seemed reasonable to assume that if a chemotherapy drug could be delivered to cancer cells in the presence of an agent which suppressed the SK1 enzyme then the combination could make for a more efficient treatment, resulting in more successful outcomes.
Professor Dmitry Pshezhetskiy and his team at UEA identified and developed a new combination cancer therapy to overcome the toxicity problem to enable the two drugs to be used in a highly targeted powerful combination.
A further enhancement for the treatment came from recent evidence showing that cancer molecular therapies can be significantly improved if the drug is attached to a nanoparticle and delivered in the form of a nanoformulation.
Based on these two observations, a new cancer chemotherapy treatment has been designed, tested, published and patented. The special nanoformulation contained two clinically approved drugs – an inhibitor to SK1, Fingolimod, together with Taxane chemotherapy. These loaded nanoparticles were designed to target the delivery of the drugs to the tumour and ‘weaken’ the cancer cells, thereby allowing Taxane to act with more efficacy
Before progressing to a full scale human trial, a series of pre-clinical studies on the patented system was carried out. This involved making a number of measurements on the nanoparticles to determine such things as drug release and the effectiveness of the new system in reducing tumour progression.
The Outcome
The new nanoparticle drug delivery system (or nanoformulation) has been further tested. The in vitro efficacy of the system was measured and found to be more effective at treating the tumour when compared to the free drugs on their own. In addition, the in vivo testing of the nanoparticle system for stability and pharmacokinetic properties was also carried out and found to perform satisfactorily.
In conclusion this project has progressed the development of a more efficient chemotherapy treatment for cancer, preparing the way for the crucial next step of clinical trial.
Next Steps
The data from this project has underpinned a clinical trial application to the MHRA for permission to carry out a Phase 1 clinical trial. Further investment is also being sought to prepare the necessary materials to carry out the trial.
The nanoparticles are also capable of delivering an imaging probe which can be used to visualise the tumour and monitor the progression of treatment. The development of this part of the system will also be explored in the future.
Full scale commercialisation of the nanoparticle drug delivery system will provide a new and more efficient chemotherapy treatment for cancer patients with advanced stages of cancer.
In the future, this innovative platform technology may also be used as a precision medicine for targeting multiple chemoresistance mechanisms in individual patients delivering tailored combinations of drugs and dosages.
Further Links
New nanoparticle cancer therapy can directly tackle cancer cells
